
This represents the formula SnF 2, which is more properly named tin(II) fluoride. For example, you may see the words stannous fluoride on a tube of toothpaste. Though this naming convention has been largely abandoned by the scientific community, it remains in use by some segments of industry. Out-of-date nomenclature used the suffixes – ic and – ous to designate metals with higher and lower charges, respectively: Iron(III) chloride, FeCl 3, was previously called ferric chloride, and iron(II) chloride, FeCl 2, was known as ferrous chloride.
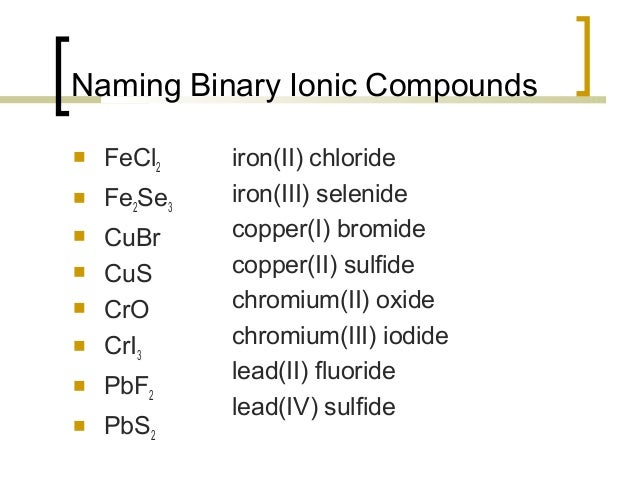
Names of Some Transition Metal Ionic Compounds These two compounds are then unambiguously named iron(II) chloride and iron(III) chloride, respectively. In cases like this, the charge of the metal ion is included as a Roman numeral in parentheses immediately following the metal name. The simplest name, “iron chloride,” will, in this case, be ambiguous, as it does not distinguish between these two compounds. Iron typically exhibits a charge of either 2+ or 3+ (see Figure 2 in Chapter 2.6 Molecular and Ionic Compounds), and the two corresponding compound formulas are FeCl 2 and FeCl 3. For example, consider binary ionic compounds of iron and chlorine. The charge of the metal ion is determined from the formula of the compound and the charge of the anion. Compounds of these metals with nonmetals are named with the same method as compounds in the first category, except the charge of the metal ion is specified by a Roman numeral in parentheses after the name of the metal. Most of the transition metals can form two or more cations with different charges. Ionic CompoundĪdded to “iodized” salt for thyroid healthīaking soda used in cooking (and as antacid)Īnti-caking agent used in powdered productsĬompounds Containing a Metal Ion with a Variable Charge Look at the label or ingredients list on the various products that you use during the next few days, and see if you run into any of those in this table, or find other ionic compounds that you could now name or write as a formula. Some of these compounds, where they are found, and what they are used for are listed in Table 9. Names of Some Polyatomic Ionic CompoundsĮvery day you encounter and use a large number of ionic compounds. NaCl, sodium chlorideĬompounds containing polyatomic ions are named similarly to those containing only monatomic ions, except there is no need to change to an – ide ending, since the suffix is already present in the name of the anion. The name of a binary compound containing monatomic ions consists of the name of the cation (the name of the metal) followed by the name of the anion (the name of the nonmetallic element with its ending replaced by the suffix – ide). First, is the compound ionic or molecular? If the compound is ionic, does the metal form ions of only one type (fixed charge) or more than one type (variable charge)? Are the ions monatomic or polyatomic? If the compound is molecular, does it contain hydrogen? If so, does it also contain oxygen? From the answers we derive, we place the compound in an appropriate category and then name it accordingly. To name an inorganic compound, we need to consider the answers to several questions. The rules for organic compounds, in which carbon is the principle element, will be treated in a later chapter on organic chemistry. We will limit our attention here to inorganic compounds, compounds that are composed principally of elements other than carbon, and will follow the nomenclature guidelines proposed by IUPAC. The simplest of these are binary compounds, those containing only two elements, but we will also consider how to name ionic compounds containing polyatomic ions, and one specific, very important class of compounds known as acids (subsequent chapters in this text will focus on these compounds in great detail). This module describes an approach that is used to name simple ionic and molecular compounds, such as NaCl, CaCO 3, and N 2O 4.

Nomenclature, a collection of rules for naming things, is important in science and in many other situations. Derive names for common types of inorganic compounds using a systematic approach.By the end of this module, you will be able to:
